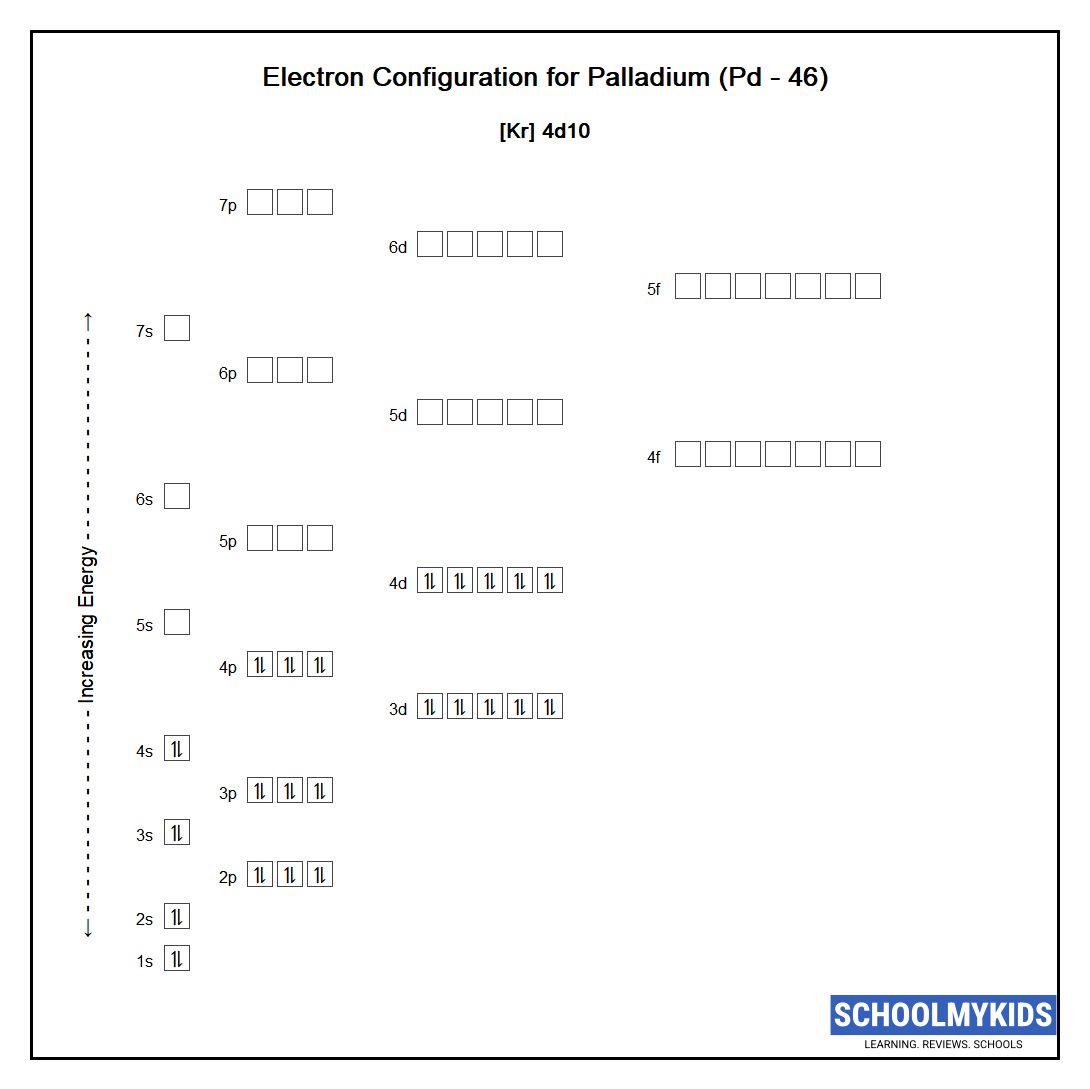
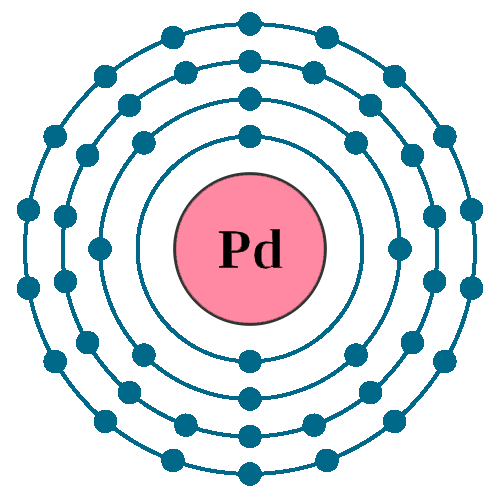
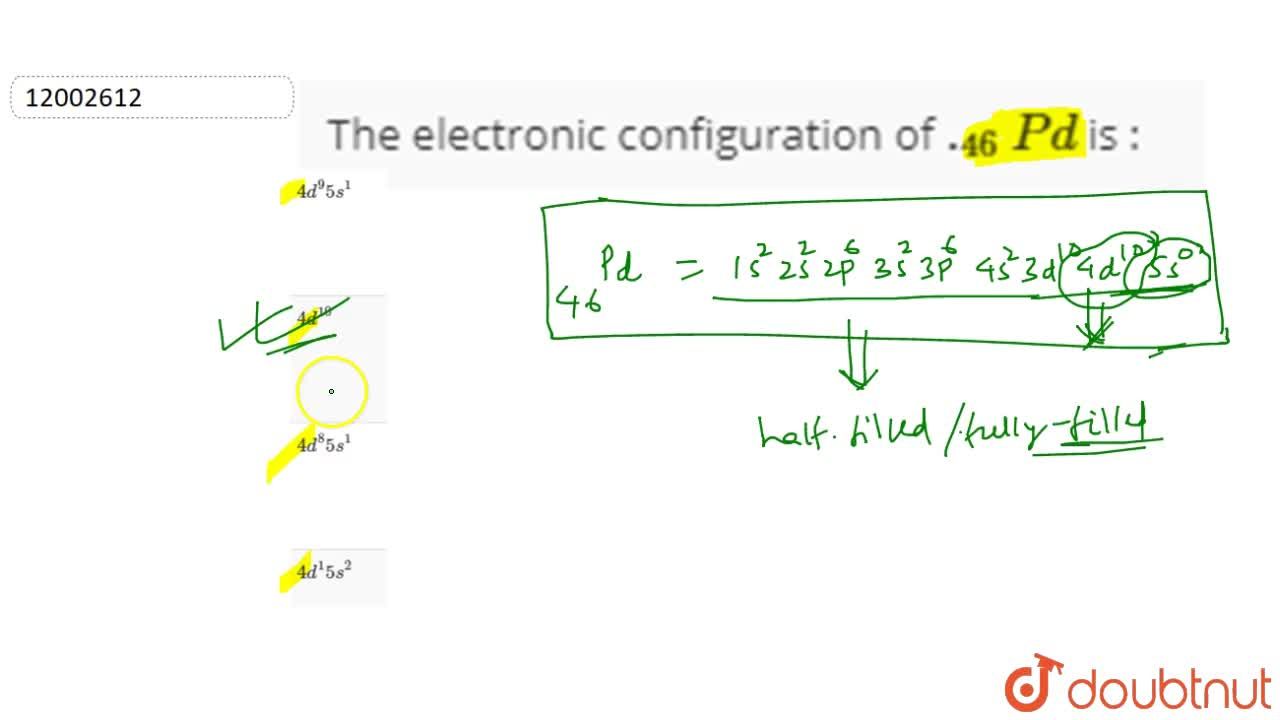
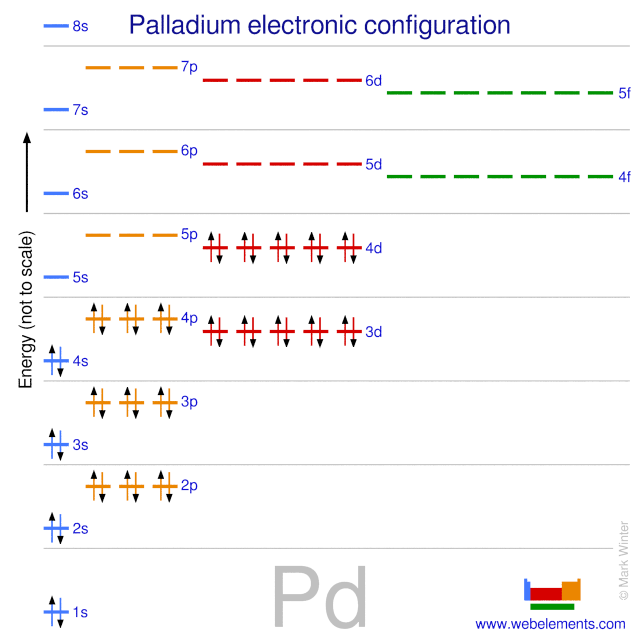
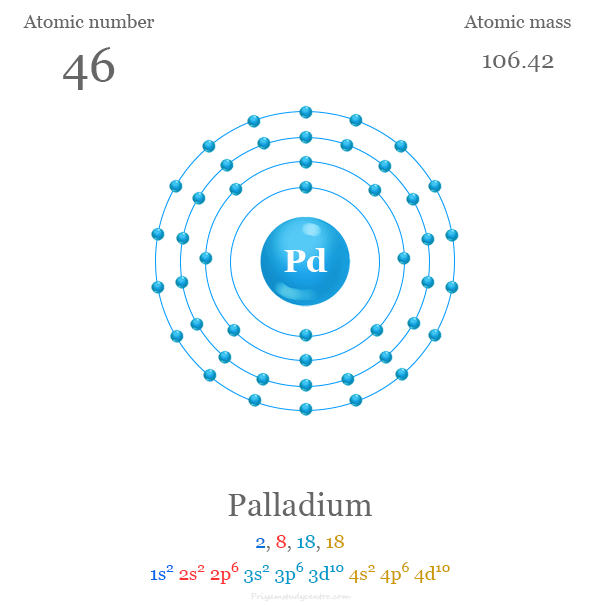
Pd has exceptional valence shell electronic configuration of `4d^(10)5s^(0)`. It is a member of- - YouTube
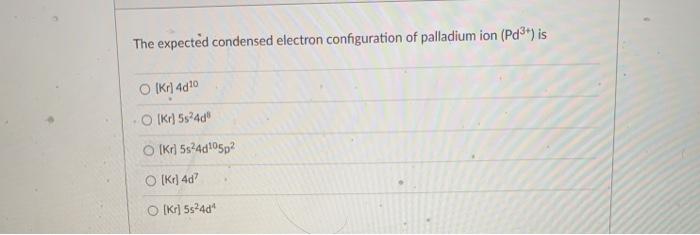
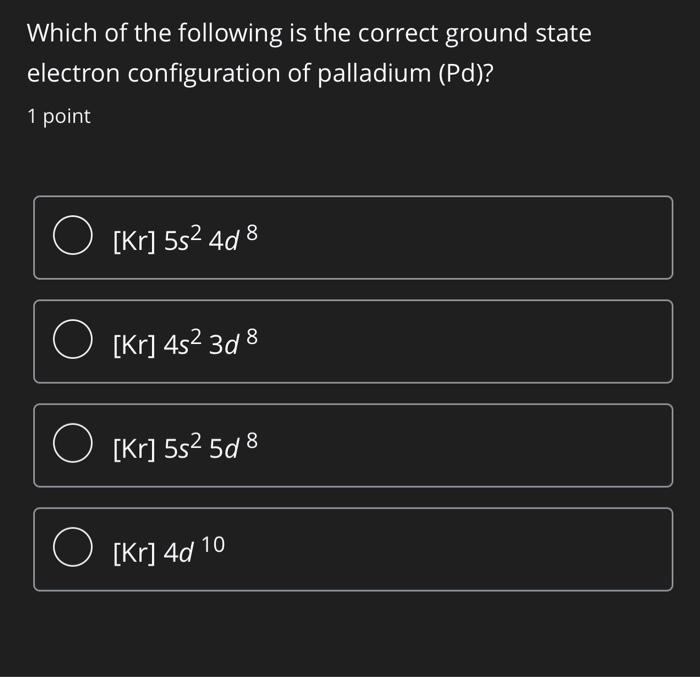
Electronic configuration of Pd (Z=46) is (1) (Kr) 4d^85s^2 (2) (Kr) 4d^9 5s^1 (3) (Kr) 4d^105s^0 (4) 4d^75s^1
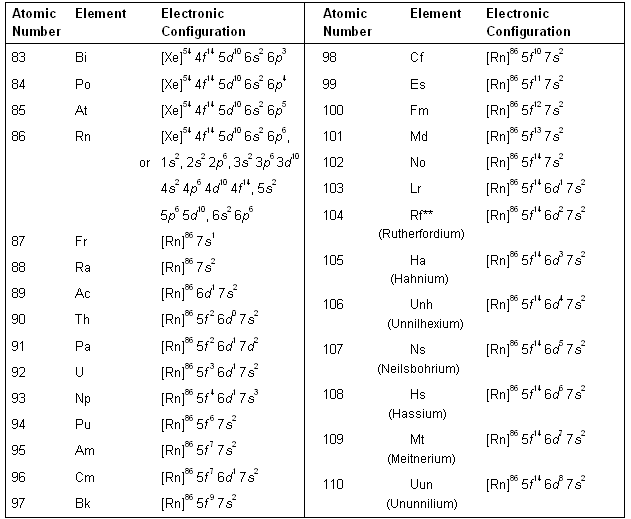
Why is palladium 5s0 4d10 more stable than 5s2 4d8, and why are the configurations of nickel and platinum different from palladium's? I had learned (and had always taught) that it is